HPTN 084-01 (The LIFT Study) is a clinical trial examining whether injectable cabotegravir (CAB) for PrEP (pre-exposure prophylaxis) is safe and acceptable for adolescent females (assigned female at birth).
HPTN 084-01 (The LIFT Study) is a clinical trial examining whether injectable cabotegravir (CAB) for PrEP (pre-exposure prophylaxis) is safe and acceptable for adolescent females (assigned female at birth).
This study will be enrolling about 50 participants at sites in South Africa, Uganda, and Zimbabwe.
 What will happen during HPTN 084-01?
What will happen during HPTN 084-01?
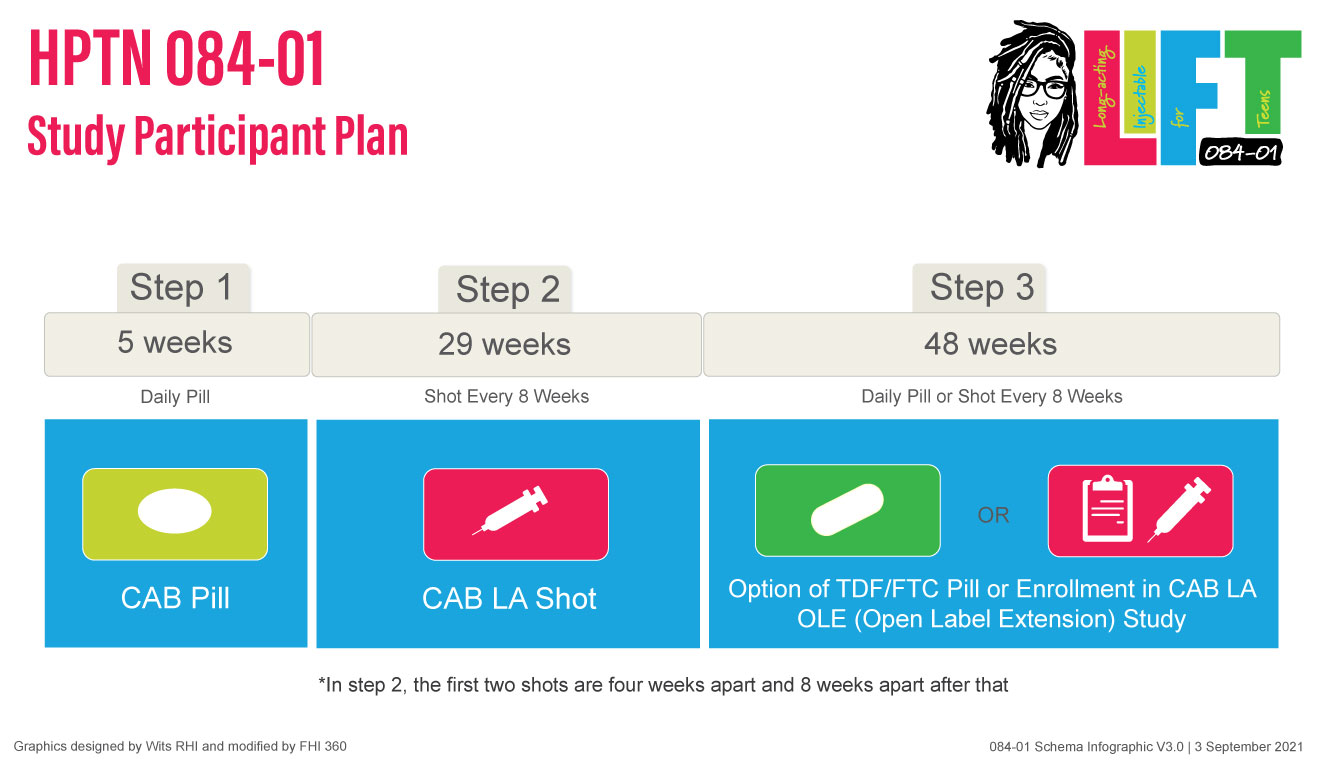
Participants will move through the study in 3 steps:
- Step 1: Participants will take one CAB pill every day for five weeks
- Step 2: Participants will receive a total of 5 CAB injections over 6 months
- Step 3: Participants will come to the clinic for study visits quarterly and have the option to take Truvada for PrEP or enroll in a CAB LA open-label extension (OLE) study for about one year.